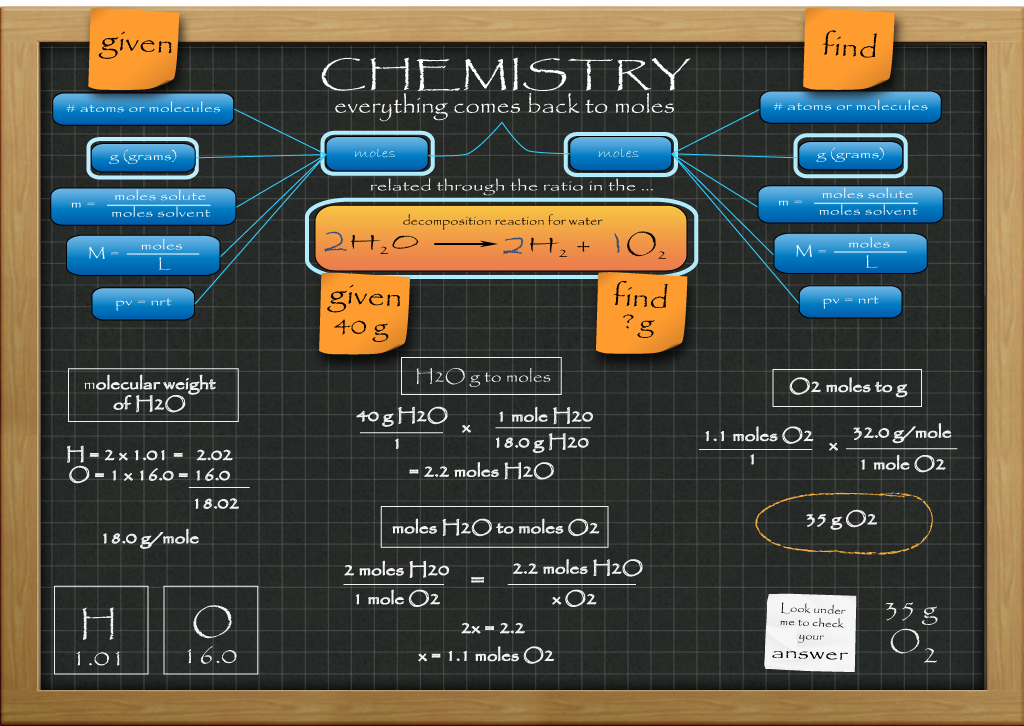
Stoichiometry
Stoichiometry is the study of the quantitative aspects of chemical reactions. It is the 'how much' side of chemistry. The image above is like a grand concept map of the quantitative relationships that will be studied this year. Everything comes back to moles there in the middle with the chemical equation that describes that particular reaction acting like a bridge that gets you from one chemical in the reaction to another chemical in the reaction. If you are given a measurement of one substance and asked for a measurement in some unit of another substance, you will have to go across this bridge to get there.
Another helpful tip is learning to thing, 'Run for the periodic table' every time you see a combination of grams and moles such as moles/gram or gram/moles. This is used whenever you will be converting between grams and moles. In the example above you can see grams has to be converted to moles to get to the middle moles to moles conversion as well as needing it again when you go from moles of the find to grams of the find.
Another helpful tip is learning to thing, 'Run for the periodic table' every time you see a combination of grams and moles such as moles/gram or gram/moles. This is used whenever you will be converting between grams and moles. In the example above you can see grams has to be converted to moles to get to the middle moles to moles conversion as well as needing it again when you go from moles of the find to grams of the find.